Hormone replacement therapy (HRT)
Conjugated Equine Oestrogen, Conjugated Equine Oestrogen with Medroxyprogesterone acetate, Conjugated Oestrogens with Bazedoxifene acetate, Estradiol, Estradiol with Dydrogesterone, Estradiol with Levonorgestrel, Estradiol with Medroxyprogesterone acetate, Estradiol with Norethisterone acetate, Estradiol with Norgestrel |
Issues for Surgery |
Risk of menopausal symptoms, e.g. hot flushes, if discontinued preoperatively. Risk of venous thromboembolism (VTE) if oral HRT continued. |
Advice in the Perioperative period |
Elective Surgery Minor surgery Major Surgery Consider other risk factors that the patient may have that further increase the risk of VTE e.g. age, weight, previous history of VTE. EXCEPT: Transdermal route – continue (see Further Information) Emergency Surgery Ensure adequate thromboprophylaxis to reduce risk of VTE1. If prolonged immobilisation anticipated consider discontinuing on admission. EXCEPT: Transdermal route – continue (see Further Information) Post-operative Advice If stopped pre-operatively restart after full mobilisation1. |
Interaction(s) with Common Anaesthetic Agents |
None1, 2. |
Interaction(s) with other Common Medicines used in the Perioperative Period |
Etoricoxib & Non-Steroidal Anti-inflammatory Drugs (NSAIDs) Etoricoxib slightly increases the exposure to conjugated oestrogens from HRT1,2. The findings of one observational study raise the possibility that the risk of myocardial infarction might be higher with the concurrent use of NSAIDs and HRT1,2. |
Further Information |
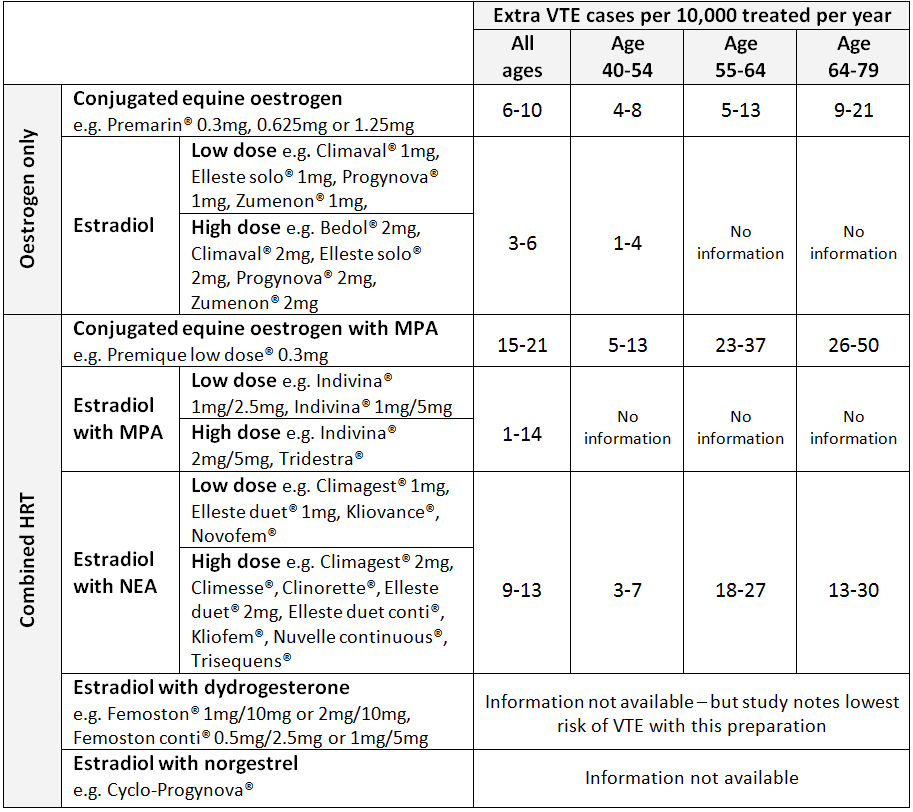
Risk of VTE with Oral Route During first pass metabolism in the liver oral HRT affects the clotting cascade by increasing resistance to protein S and protein C (natural anticoagulants) and increasing fibrinogen, thus increasing the risk of thrombosis3. Patients taking oral HRT are at increased risk of VTE compared with non-users3. The risk is thought to be greatest in the first year of treatment and also in women with an increased body mass index3. The baseline risk of VTE also increases substantially with age1, 3, 4. The risk of VTE varies depending on the oestrogen (and progesterone) content of the preparation (see figure 1)3, 4. Preparations with conjugated equine oestrogen were associated with higher VTE risk compared to those with estradiol4. Higher doses of oestrogen were also associated with higher VTE risk4. The highest risk is with conjugated equine oestrogen and medroxyprogesterone acetate and the lowest risk is with estradiol with dydrogesterone4.
Figure 1 - Extra VTE cases per 10000 treated per year (95% confidence interval) for different HRT preparations compiled from UK population data. MPA = medroxyprogesterone acetate, NEA = norethisterone acetate1, 4 Risk of VTE with Transdermal Route Transdermal administration of oestrogen avoids first pass metabolism and thus has less effect on the coagulation factors than oral HRT3. The risk associated with transdermal HRT given at standard therapeutic doses is no greater than the baseline population risk3, 4. Since transdermal HRT has little or no impact on coagulation and is not associated with an increased risk of VTE it doesn’t need to be discontinued before elective surgery3.
|
References |
|